
Thus, the formula for carbon dioxide is CO 2, since the name indicates one carbon (no prefix), and two oxygens (prefix di- means 2). Apply the prefix when writing the formula. The number of each element in the compound is indicated by a prefix. Writing the formula of a binary molecular compound is actually quite easy. Writing Formulae of Binary Molecular Compounds This means that two iron(III) ions, totaling +6, and three carbonate ions, totaling -6 are needed to write the formula: Fe 2(CO 3) 3. One needs to consider a common multiple, in this case 6, and adjust the number of each ion to this multiple. The magnitudes of the charges are not equal, and they are not multiples of each other. Carbonate ions has the formula, CO 3 2-, and thus has a -2 charge. Iron(III) has a +3 charge, as indicated by its Stock number. What is the formula for iron(III) carbonate?Ĭonsider the ions. Here is a compound which is a little more difficult. In order to show three nitrate ions in the formula, one must use parenthesis around the nitrate ion and a subscript of 3 outside the parenthesis: Cr(NO 3) 3. Three nitrate ions at -1 apiece are needed to cancel the +3 on the chromium ion. The magnitude of the chromium(III) ion is greater than the magnitude of the nitrate ion. Nitrate ion you have memorized to be NO 3 -, and has a -1 charge. The Roman numeral, III, for chromium(III), indicates the charge of the chromium ion is +3. What is the formula for the compound, chromium(III) nitrate?Ĭonsider th ions. Let's consider a compound involving a Stock number. 
Since two sodium ions are needed, a subscript of 2 will be found in the formula: Na 2SO 4. One can see that more than one sodium ion will be needed to cancel the -2 charge on the sulfate ion. The magnitude of the sulfate ion is greater than the magnitude of the sodium ion. Sulfate ion is SO 4 2-, and has a -2 charge. Sodium, found in GroupIA, can only form a +1 ion. What is the formula for the compound, sodium sulfate?Ĭonsider the ions. Polyatomic anions, with few exceptions, will have names ending in -ite or -ate, so they should be easy to recognize. Let's consider a compound containing a polyatomic anion. Incidently, by convention, the cation is always written first: AlCl 3. To show three chloride ions in the formula a subscript of three is used. In fact, three chloride ions at -1 apiece are needed to cancel the +3 on aluminum ion. One can see that more than one chloride ion will be needed to cancel the +3 charge on aluminum. The magnitude of the aluminum ion is greater than the magnitude of chloride ion. Chloride ion must have a -1 charge, because it is found in GroupVIIA. Aluminum, found in GroupIIIA, can only form a +3 ion. What happens when the magnitude of the charges are not the same? For instance, what will be the formula of the compound, aluminum chloride?Īs before, consider the ions involved. Since the magnitude of the charges are the same, the ions will combine in a 1:1 ratio, and the formula for magnesium oxide is MgO. Oxide ion must have a charge of -2, because oxygen is found in GroupVIA. 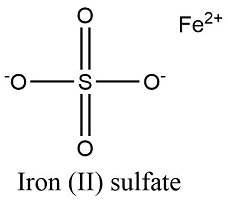
Magnesium, found in GroupIIA, can only form a +2 ion. What is the chemical formula for the compound, magnesium oxide?įirst, consider the ions involved. Let's look at a few examples, and try to come up with the chemical formulae. For the monatomic anions, the charge is equivalent to the Group number - 8. Cations which can form more than one ion will have a Stock number shown as a Roman numeral in parenthesis. Recall that some cations will only form one ion (GroupIA, IIA, and IIIA, except Tl). If you haven't memorized the ions, it is essential you do so immediately. The inability to recognize these ions are the main reason for difficulty in writing chemical formulae of inorganic compounds. These include the cations, as well as the monatomic anions and polyatomic anions. You must recognize the ions which are a part of the compound. For example, potassium oxide is made up of potassium ions K + atom.When writing a chemical formulae from the name of an ionic compound, one must consider the ions contained in the compound.(If it had a different charge, it would pull in another atom.) Find the number of atoms of each element that combine to "cancel out" each other's charge and make a neutral compound. Taken as a whole, the ionic compound is electrically neutral, meaning it has a total charge of zero. Electrical forces hold together ionic compounds, pulling the positive and negative ions together. It’s crucial to balance the charges in order to write ionic compounds correctly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
